Understanding the pH Scale: A Complete Information to Acids, Bases, and Their Significance
Associated Articles: Understanding the pH Scale: A Complete Information to Acids, Bases, and Their Significance
Introduction
With nice pleasure, we are going to discover the intriguing subject associated to Understanding the pH Scale: A Complete Information to Acids, Bases, and Their Significance. Let’s weave attention-grabbing info and supply contemporary views to the readers.
Desk of Content material
Understanding the pH Scale: A Complete Information to Acids, Bases, and Their Significance
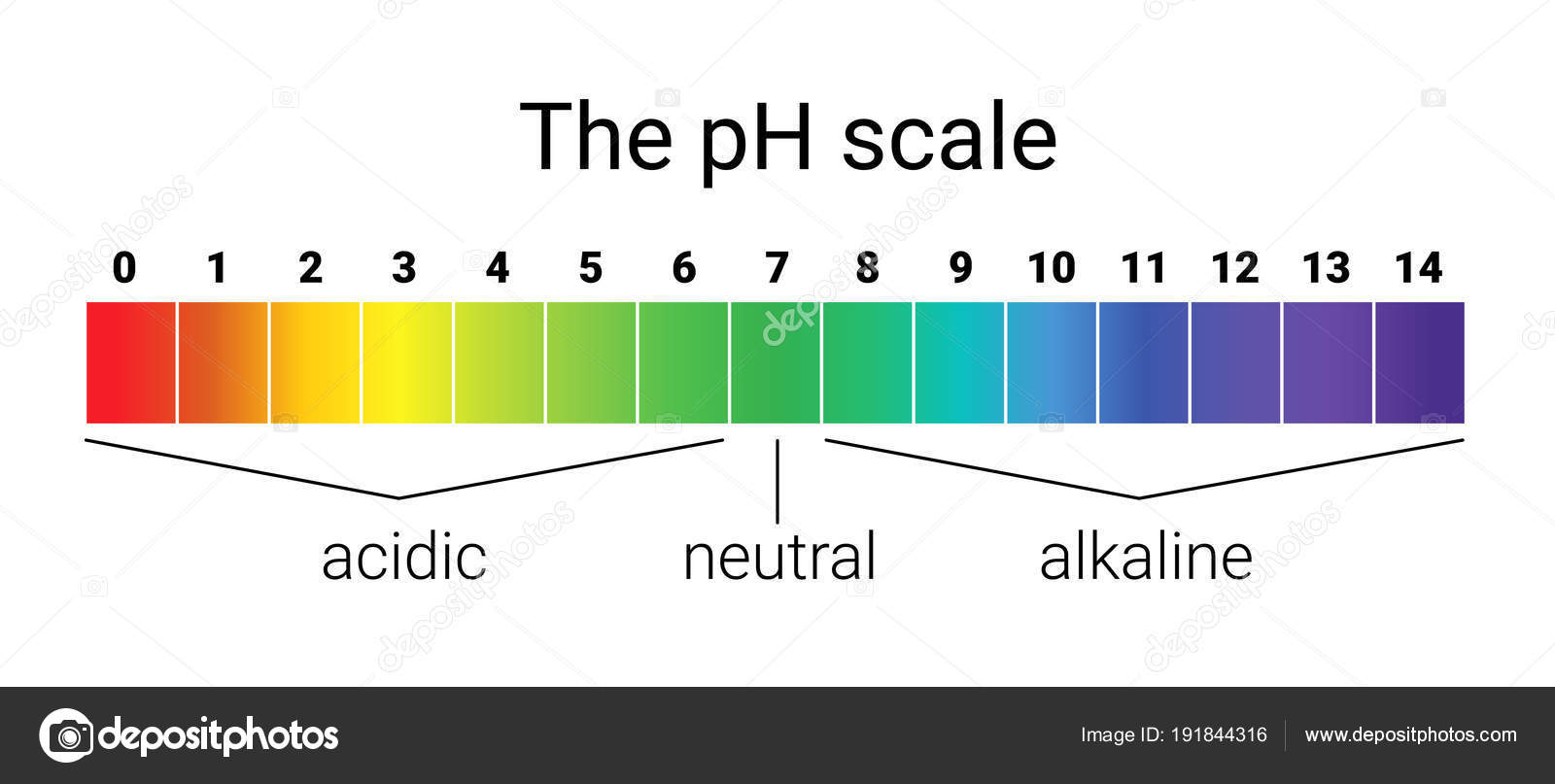
The pH scale is a basic idea in chemistry and has far-reaching implications throughout varied fields, from biology and medication to environmental science and industrial processes. It quantifies the acidity or basicity (alkalinity) of an answer, offering an important metric for understanding and controlling chemical reactions and organic processes. This text delves into the intricacies of the pH scale, exploring its logarithmic nature, the distinctions between acids and bases, the importance of impartial pH, and the purposes of pH measurement in numerous contexts. We can even present an in depth pH chart illustrating the acidity and basicity of frequent substances.
The Nature of the pH Scale:
The pH scale is a logarithmic scale starting from 0 to 14, representing the focus of hydrogen ions (H⁺) in an answer. A decrease pH worth signifies a better focus of H⁺ ions, signifying a extra acidic resolution. Conversely, a better pH worth signifies a decrease focus of H⁺ ions, signifying a extra fundamental (alkaline) resolution. The dimensions is logarithmic, that means that every complete quantity change represents a tenfold change in H⁺ ion focus. As an illustration, an answer with a pH of three is ten occasions extra acidic than an answer with a pH of 4, and 100 occasions extra acidic than an answer with a pH of 5.
The pH of an answer is calculated utilizing the next system:
pH = -log₁₀[H⁺]
the place [H⁺] represents the molar focus of hydrogen ions.
Acids and Bases: A Definition:
The ideas of acids and bases have been refined over time, resulting in a number of totally different definitions. Essentially the most generally used are the Arrhenius, Brønsted-Lowry, and Lewis definitions.
-
Arrhenius Definition: That is the best definition, stating that an acid is a substance that produces hydrogen ions (H⁺) when dissolved in water, whereas a base is a substance that produces hydroxide ions (OH⁻) when dissolved in water. This definition, nevertheless, is proscribed because it solely applies to aqueous options.
-
Brønsted-Lowry Definition: This definition is broader and defines an acid as a proton (H⁺) donor and a base as a proton acceptor. This definition encompasses a wider vary of gear than the Arrhenius definition and does not require the presence of water.
-
Lewis Definition: That is essentially the most normal definition, defining an acid as an electron-pair acceptor and a base as an electron-pair donor. This definition encompasses much more substances than the Brønsted-Lowry definition, extending the idea of acids and bases past proton switch.
Impartial pH:
A impartial pH is outlined as 7 at 25°C (77°F). At this pH, the focus of hydrogen ions (H⁺) is the same as the focus of hydroxide ions (OH⁻). Pure water, at 25°C, has a pH of seven. Nonetheless, it is essential to notice that the pH of pure water can differ barely with temperature.
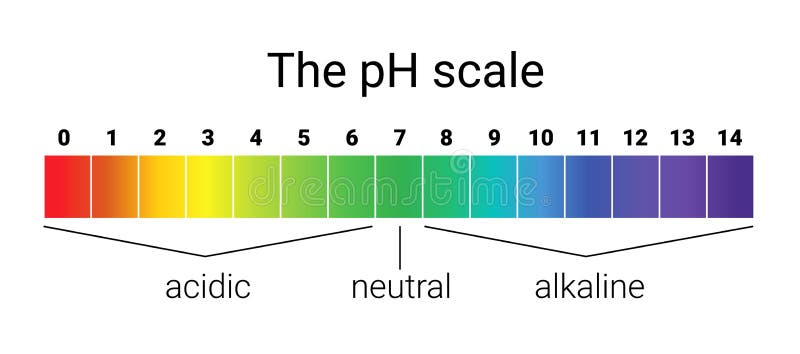
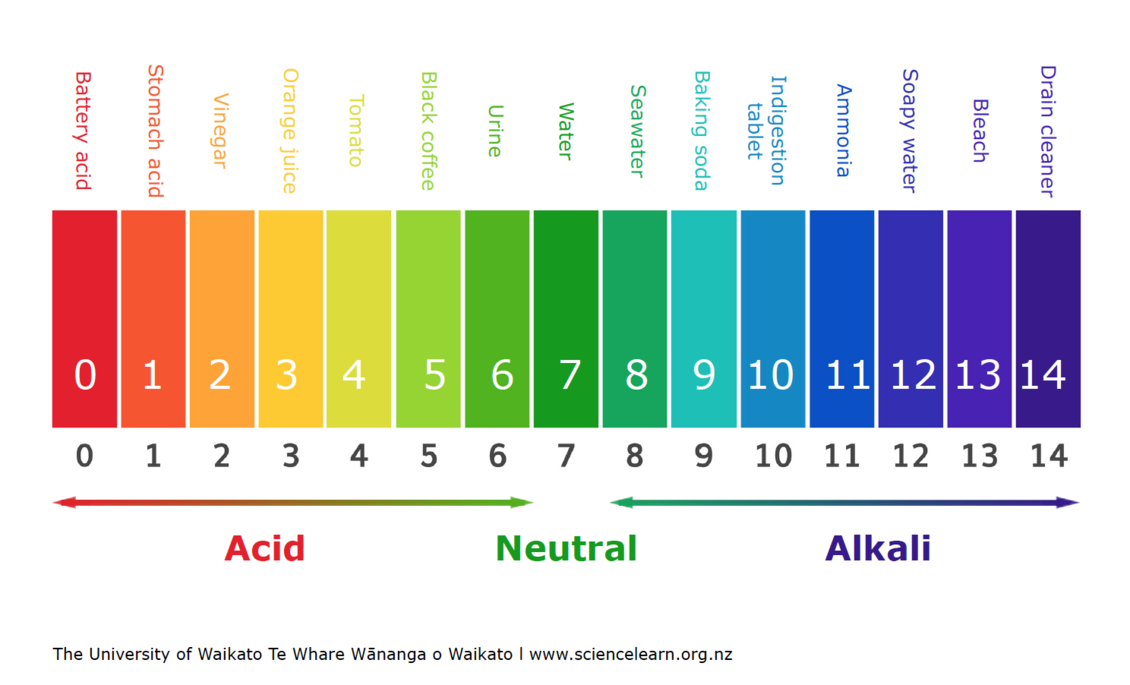
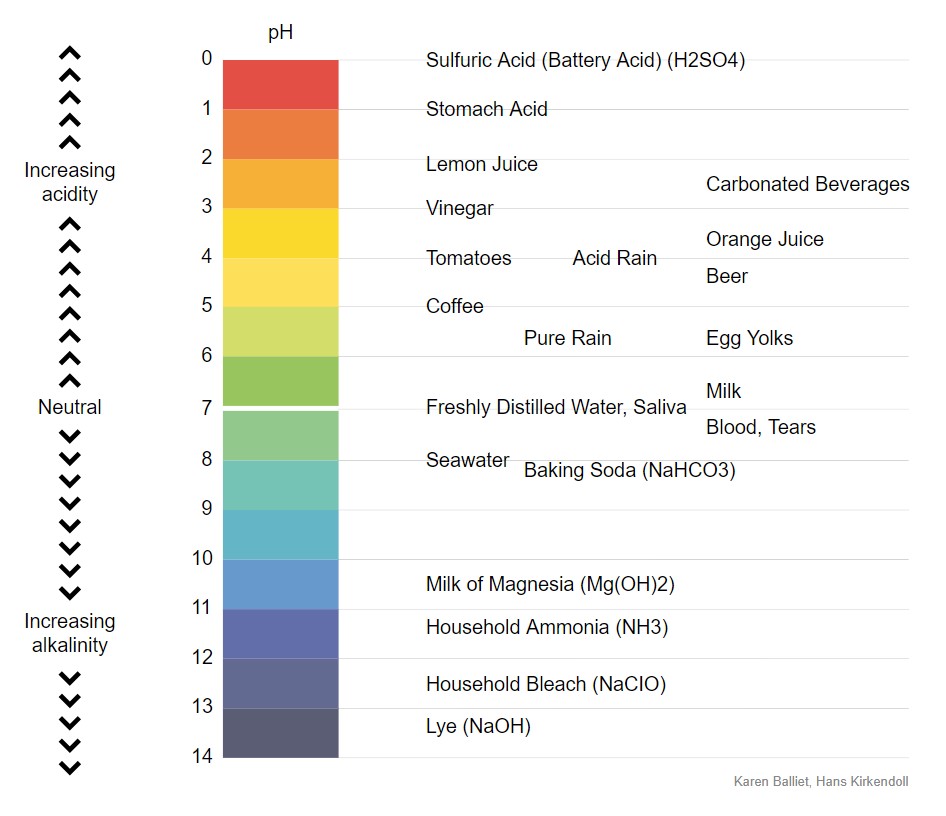
The pH Chart: A Visible Illustration of Acidity and Basicity:
The next chart supplies a visible illustration of the pH scale and the relative acidity and basicity of frequent substances:
| pH Degree | Description | Examples |
|---|---|---|
| 0-2 | Sturdy Acid | Battery acid, abdomen acid, hydrochloric acid |
| 2-4 | Weak Acid | Vinegar, lemon juice, orange juice, cola |
| 4-6 | Reasonably Acidic | Rainwater, espresso, black tea |
| 6-7 | Barely Acidic | Urine, saliva |
| 7 | Impartial | Pure water |
| 7-8 | Barely Alkaline | Blood, seawater |
| 8-10 | Reasonably Alkaline | Baking soda resolution, seawater |
| 10-12 | Weak Alkaline | Milk of magnesia, cleaning soap |
| 12-14 | Sturdy Alkaline | Drain cleaner, ammonia |
Vital Concerns:
-
Temperature Dependence: The pH of an answer could be affected by temperature. Modifications in temperature can alter the ionization of water and the dissociation of acids and bases.
-
Focus Dependence: The pH of an answer is straight associated to the focus of the acid or base. A extra concentrated resolution will typically have a extra excessive pH (both extra acidic or extra fundamental).
-
Buffer Options: Buffer options are options that resist adjustments in pH upon the addition of small quantities of acid or base. They’re essential in sustaining secure pH ranges in organic techniques and chemical processes.
Functions of pH Measurement:
The measurement of pH is crucial throughout a variety of purposes:
-
Environmental Monitoring: pH measurement is essential for assessing water high quality, monitoring soil situations, and monitoring air air pollution. Acid rain, for example, is characterised by a lower-than-normal pH.
-
Agriculture: Soil pH is a important issue affecting plant progress. Totally different crops thrive in numerous pH ranges, and soil pH must be adjusted to optimize crop yields.
-
Drugs and Healthcare: Blood pH is fastidiously regulated inside a slim vary (7.35-7.45) to keep up correct bodily capabilities. Deviations from this vary can result in severe well being issues. pH measurement can be utilized in varied diagnostic checks and medical remedies.
-
Meals and Beverage Business: pH management is important in meals processing and preservation. It impacts the style, texture, and shelf lifetime of meals merchandise. It additionally performs an important position in fermentation processes.
-
Industrial Processes: Many industrial processes require exact pH management to make sure environment friendly and protected operation. This contains chemical manufacturing, wastewater remedy, and varied different purposes.
-
Aquatic Techniques: Sustaining the suitable pH in aquariums and different aquatic techniques is essential for the well being and survival of aquatic organisms.
Measuring pH:
pH is often measured utilizing a pH meter, a tool that makes use of a pH-sensitive electrode to measure the voltage distinction between the electrode and a reference electrode. The voltage distinction is then transformed right into a pH studying. Different strategies embrace the usage of pH indicator options or pH take a look at strips, which change coloration relying on the pH of the answer. These strategies are much less exact than pH meters however are sometimes extra handy and cost-effective for much less demanding purposes.
Conclusion:
The pH scale is a strong instrument for understanding and controlling the acidity and basicity of options. Its significance extends throughout varied scientific disciplines and industrial purposes. Understanding the rules of the pH scale, the distinctions between acids and bases, and the strategies of pH measurement is crucial for anybody working in fields the place chemical reactions and organic processes play a major position. The cautious monitoring and management of pH are essential for sustaining environmental well being, guaranteeing the protection and effectiveness of business processes, and safeguarding human well being. Continued analysis and improvement in pH measurement applied sciences will undoubtedly result in additional developments in varied fields, reinforcing the enduring significance of this basic idea in chemistry.





Closure
Thus, we hope this text has supplied useful insights into Understanding the pH Scale: A Complete Information to Acids, Bases, and Their Significance. We hope you discover this text informative and useful. See you in our subsequent article!